Mastery Across All Drug Modalities
From discovery to development, we bring deep expertise in every modern therapeutic approach to accelerate your drug development journey.
Small Molecules
Orally bioavailable compounds targeting intracellular pathways with high specificity and tunable pharmacokinetics.
Biologics
Large, complex proteins like monoclonal antibodies designed for high-affinity targeting of disease mechanisms.
Peptide Therapeutics
Mid-sized molecules bridging the gap between small molecules and biologics, offering high specificity.
Cell & Gene Therapy
Revolutionary treatments that modify or replace genetic material to treat disease at its root cause.
Oligonucleotides
Short nucleic acid chains that modulate gene expression with exquisite precision, enabling targeting of 'undruggable' pathways.
Antibody-Drug Conjugates (ADCs)
Combining monoclonal antibodies with potent cytotoxic payloads to deliver chemotherapy directly to diseased cells.
Our Competitive Edge
We combine deep scientific expertise with cutting-edge technology to deliver unparalleled value and accelerate your journey to market.
Regulatory Insight
Proactive guidance to navigate complex global compliance landscapes with confidence.
AI-Powered Efficiency
Leveraging machine learning to optimize processes and reduce development timelines.
End-to-End Platform
An integrated service from discovery to GMP manufacturing.
Strategic GMP & ICH Implementation — Right From the First Time
Molgenium embeds GMP and ICH compliance into your process DNA — not as an afterthought, but as a strategic foundation. We ensure your systems are audit-ready, scalable, and scientifically sound — from Day One.
Gap Analysis & Roadmap
We assess your state against ICH Q7-Q10 & EU/FDA GMP, building a prioritized, risk-based roadmap aligned with your goals.
Design Quality into Systems
We co-design documentation and workflows using QbD principles, ensuring compliance is built-in.
Train & Embed Culture
Role-based GMP/ICH training with real-world scenarios ensures your team owns compliance as a core value.
Document & Digitize
We implement lean, audit-proof documentation systems (SOPs, logs) with version control to reduce errors.
Audit Simulation & CAPA
We stress-test your system with mock audits and build robust CAPA workflows, turning findings into improvements.
Sustain & Scale
We embed metrics and reviews to ensure your GMP system evolves and stays inspection-ready.
Our Manufacturing Validation Process
Our validation doesn't just check boxes — it builds commercial confidence and de-risks your entire development pathway.

Protocol Design & Risk Mapping
We co-develop science-driven validation protocols with risk-based approaches (ICH Q9) to focus on critical process parameters that impact quality.

Installation & Operational Qualification (IQ/OQ)
We verify equipment and systems are installed correctly and operate within defined ranges — ensuring reproducibility from Day 1.

Performance Qualification (PQ)
We execute real-world runs under commercial conditions to prove consistency, yield, purity, and compliance — batch after batch.

Documentation & Regulatory Package
We deliver audit-ready documentation: protocols, reports, deviations, and traceability — aligned with FDA/EMA/ICH guidelines.
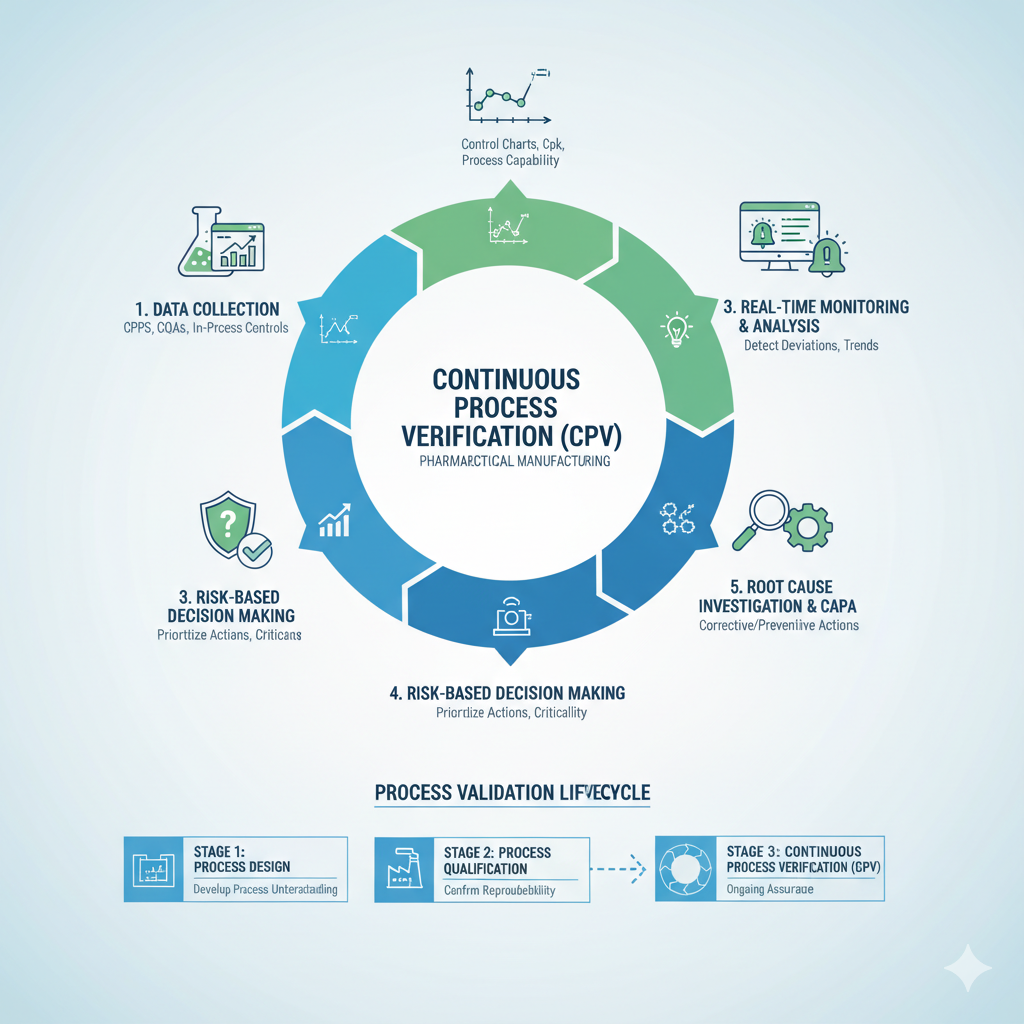
Continuous Process Verification (CPV)
We implement real-time monitoring and statistical control to ensure your process remains in a state of control — even post-approval.
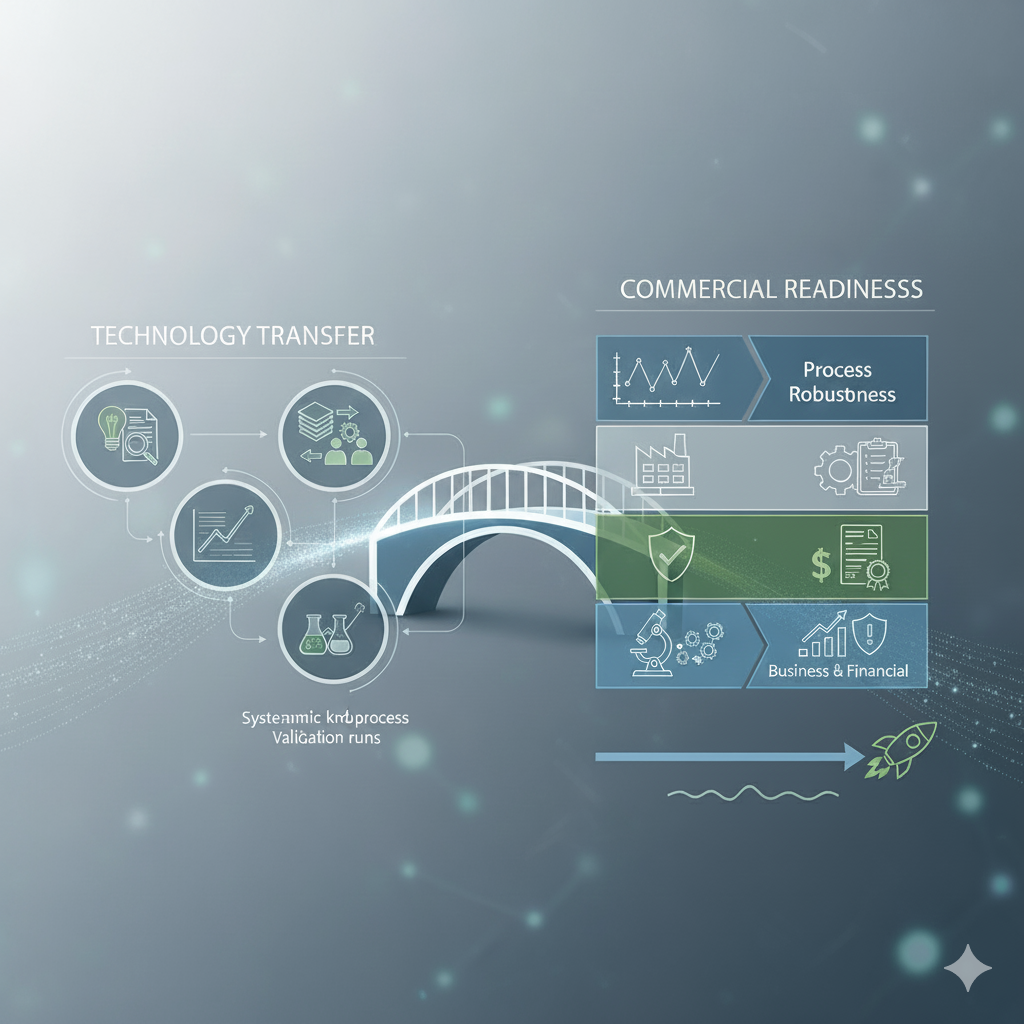
Commercial Readiness & Tech Transfer
We prepare your validated process for seamless tech transfer to internal or external manufacturing sites — accelerating time-to-market.
The Molgenium Validation Impact
We don’t just validate processes — we de-risk your entire development pathway and unlock commercial value.
0% Faster Timelines
Average reduction by avoiding rework with our first-time-right validation strategies.
0+ Passed Inspections
Our documentation has a proven track record in regulatory inspections.
Up to 0% Cost Reduction
Focus resources on what matters — reduce the cost of poor quality significantly.
A Synergistic Partnership for mRNA Therapeutics
Molgenium and Novoprotein have established a powerful collaboration, creating a one-stop CDMO service platform to accelerate development from concept to commercialization.
MOLGENIUM + NOVOPROTEIN
Combined Expertise, Accelerated Results
Our strategic partnership establishes a seamless path from non-clinical research to IND application, leveraging Molgenium's RNA synthesis platform and Novoprotein's enzyme technology.
- End-to-end mRNA process development
- Analytical method development & validation
- Research-grade mRNA production at scale
Research
Joint discovery and innovation
Development
Therapeutic candidate optimization
Production
GMP manufacturing at scale
Delivery
Commercial product distribution
A Flexible Platform for Modern Pharma
Our system is built on a foundation of core capabilities designed for compliance, efficiency, and scalability, adapting seamlessly to your unique workflow.
AI-Powered Process Optimization
Speeds up routine tasks while ensuring full traceability and data integrity.
Modular & Compliant System
Each module aligns with ICH, EMA, FDA, and ISO standards for global compliance.
Built-in Risk Management
Integrates risk assessment (ICH Q9) into every stage for continuous improvement.
Adapts to Your Workflow
Paper-Based
Full support for traditional documentation processes.
Hybrid
Seamlessly bridge mixed digital and paper systems.
Fully Digital
Leverage end-to-end automated workflows for max efficiency.
Get in Touch
We'd love to hear from you. Send us a message and we'll respond as soon as possible.
Contact Information
Email Us
info@molgenium.comBusiness Hours
Monday - Friday: 9:00 AM - 5:00 PM CET
